JPR Index | Virtual Library | Magazine Rack | Search
By Robin Whyatt
Young children generally receive greater exposure to pesticides in food than adults. Relative to their weight children both consume more food than adults (due to a higher rate of metabolism) and larger quantities of fruit (commodities that have a high likelihood of being contaminated with pesticides). A study by the Natural Resources Defense Council (NRDC)t found that for almost all of the 23 pesticides evaluated, preschoolers received greater pesticide exposure than adults. For the majority, their exposure was four or more times greater and for some pesticides as much as nine to twelve times greater.
Young children not only receive greater pesticide exposure, they may also be more susceptible to the toxic effects of these pesticides as a result of their immature physiological development. While relatively few pesticides have been specifically evaluated to determine if their toxicity is greater in the young, numerous studies have found the young to be more vulnerable to the toxic effects of many chemicals, including some pesticides.
Experimental studies have found that neurotoxic organophosphate pesticides are more acutely toxic to the young.
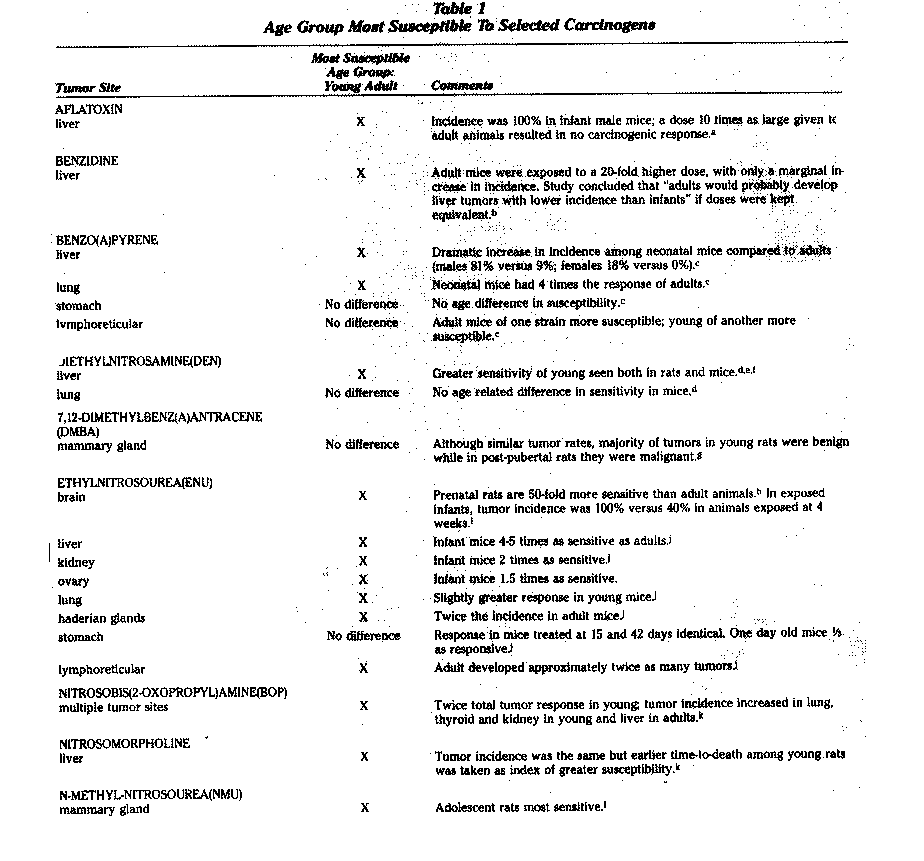
Several studies suggest that low-level exposure to some organophosphates during nervous system development may cause long-term neurological impairments. Experimental studies have also found the young to be particularly vulnerable to the effects of many carcinogens (Table 1).
Most of the evidence for the increased sensitivity in the young has come from experimental studies in laboratory animals. Human data are generally not available; however, results from experimental studies in animals provide a reasonable model to predict results in humans.2
This susceptibility of the young to carcinogens and neurotoxins is of concern in light of the preschooler's greater exposure to pesticide residues in food, many of which are carcinogenic or neurotoxic "
In general, experimental studies have found the young of various species more likely to retain a greater portion of a given dose of certain toxins because of increased absorption and decreased elimination. Further, young bodies are not as capable of segregating toxins (i.e., keeping toxins from the target organs), and the target organs themselves may be more susceptible to the toxic action of compounds.
The enhanced retention results, in part, from the fact that the gastrointestinal tract is more permeable in the young and the process by which compounds are actively transported across cell membranes is increased.3 A number of compounds particularly in organic chemicals like lead and mercury, are absorbed more readily by the young than by the adult. Developing kidneys are also less capable of excreting certain compounds in infancy than during adulthood.4
Greater retention also occurs because detoxifying enzymes are not fully functional at birth; the young, therefore, are less capable of detoxifying many chemicals than adults.5 In rodents, most detoxifying enzymes are found at very low levels at birth, with marked elevations occurring at the time of weaning.6 In human infants, there is appreciable enzyme activity, but enzyme levels are still lower than later in childhood or during adulthood.6 Consequently, chemicals can be retained in the blood of newborns for longer periods than they are in adults.5 The greater neurotoxicity of the pesticides methyl parathion and parathion in young rats has been shown to be the result of the low levels of detoxifying enzymes during this age period.'
Besides having a propensity for retaining a greater portion of a given compound, the young are less capable of binding many compounds to proteins in the blood.3 Protein binding can be a significant detoxification mechanism, since it can segregate a chemical foreign to the body, making it less likely that the compound will reach its site of toxic action.
Further, the target organs themselves, because of physiological immaturities, can be more susceptible to the toxic action of a chemical. For instance, the brain of most species is not fully developed for a long period following birth.4 It is thought that this protracted period of maturation contributes to the sensitivity of the developing brain to neurotoxins.3
Are the young always at increased risk? The answer is no. Age-related susceptibility is a continuum and the young may be more sensitive, equally sensitive, or less sensitive to specific chemicals than adults.5 In some instances, rather than increasing susceptibility, immaturities in the physiological system actually protect against toxic insults. For example, the immature kidney is often less susceptible to the effects of toxic chemicals than the adult kidney.3 The young are generally more resistant to kidney toxins as well as to certain respiratory and river toxins than adults.3
However, the young appear to be more susceptible than adults to the lethal effects of many compounds. A recent review of 269 drugs and toxic substances, including a number of pesticides, found that the lethal dose (LD5o in mg/kg) was longer in newborn rodents than in adult rodents in 86% of the cases.3 In 17% of the cases where the lethal dose was longer, it was more than 10 times longer.
Further, the young have been found to be more susceptible to the effects of many carcinogens and neurotoxins.3 This susceptibility to carcinogens and neurotoxins is of concern in light of the preschooler's greater exposure to pesticide residues in food, many of which are carcinogenic or neurotoxic.
A number of laboratory studies have found that animals are at greater risk of developing cancer if exposure began during infancy rather than later in life. Of 14 carcinogens reviewed by NRDC (Table 1), the young were more susceptible to 12. Similarly, another recent review found the young more susceptible to eight out of ten carcinogens evaluated.3
While there is relatively little human experience to draw on, children are more susceptible to radiation exposure, and cancers develop at most sites with greater frequency if exposure begins during childhood rather than during adulthood.9 These and other findings have led researchers to conclude that infancy has "proved to be the most susceptible period to carcinogenesis."'°
Exposure to carcinogens in infancy and early childhood does not necessarily mean that cancer will result during childhood. In fact, in most of the laboratory studies, cancers appeared late in life, regardless of whether the exposure was begun in infancy or in early adulthood. However, the cancer incidence was greater in the infant exposed groups. Infant exposure can also result in a shortening of the latency period (the time it takes for the tumor to develop) but generally the cancer is still manifested in adulthood, not during childhood. For example, in the human population, most tumors following radiation exposure during child hood do not appear until adulthood.9
One reason that the young may be more susceptible than adults to carcinogens is because cells are dividing more rapidly during childhood than later in life. Carcinogenesis is a multistage process. The first stage is known as initiation. Typically, a cell is "initiated" when a carcinogen interacts with its genetic material (DNA), causing a mutation. If the cell divides before the mutation can be repaired, the daughter cell permanently contains altered DNA. In other words, the mutation is "fixed." If the cells are dividing rapidly following exposure to a carcinogen capable of mutating DNA, there is greater probability that the mutation in DNA will be fixed and the carcinogenic event initiated.* A number of studies have correlated rapid cell division with increased cancer incidence."-'4
Another reason that the young may be more susceptible to carcinogens than adults is simply because they have a much longer life over which the carcinogenic action may occur. Following cancer initiation, subsequent events, known as promotion and progression, can cause the initiated cell to become malignant. However this usually takes place over several decades and the fully developed malignancy does not occur until much later in- life. Therefore, cells that are initiated fate in human life, i.e., after age 50, have little chance of being promoted over the remaining life of the individual and usually will not lead to cancer. However, cells that are initiated in childhood have a much higher probability of being promoted over the seventy or more years of expected life of the individual and, thus, of advancing to cancer. In fact, most of the cancer risk that an individual faces from environmental carcinogens, such as from carcinogenic residues n foods, may be from exposure in the very first years of life.'
In addition to the increased susceptibility of the young to carcinogens, age-related susceptibility has been documented for a number of neurotoxins. Of the 31 neurotoxic metals, pesticides and other organics analyzed in one review, there was an age-related difference in susceptibility for all but two. In 66% of the cases where susceptibility differed with age, the young were more susceptible.3 The young have been found to be at greater risk to the neurotoxic effects of many metals, a large number of anesthetics and analgesics, and a variety of other neurotoxic agents. For example, the young of many species, including humans, are extremely susceptible to the neurotoxic effects of lead, while: adults are fairly resistant.3' 8' 15
The young have also been shown to be at great risk to the neurotoxic effects of a number of pesticides.3 For example, the young are especially susceptible to the acute effects of the organophosphate insecticides, which disrupt normal nerve transmission. In fact, young rats are more susceptible than adults to the lethal effects of 15 out of 16 organophosphate insecticides tested.3 For parathion and methyl parathion, the fatal dose in the young rat is six to eight times longer in relation to body weight than in adult rats.7 For some organophosphate compounds, the fatal dose in immature animals has been reported to be only 1% of the lethal dose in adult animals.
The sensitivity of the young to certain neurotoxic agents is caused, in part, by the fact that the blood-brain barrier, or the barrier that inhibits the passage of compounds from the blood into the brain, is not fully developed. For example, the greater neurotoxicity of morphine, lead, and mercury in the young is related to the greater accumulation of these substances in the immature brain.3 "
The brain is also more susceptible to certain compounds during the developmental step called myelination, the process by which nerve fibers are covered by a fat-like substance (called myelin). Myelination takes place primarily after birth. For example, in humans, the most rapid phase of myelination ends at two years. However, total myelination is not complete until adolescence.4 ~s Hexachlorophene, a fungicide and antibacterial agent, is a compound that damages myelin and is particularly neurotoxic to the young, including human infants.'5, 17' 16 In France, about 40 infant deaths resulted in 1972 when baby powder, accidentally contaminated with 6.6% hexachlorophene, was used on the infants." The young are also more sensitive to certain neurotoxins, in part, because the blood capillary system in the brain is not fully developed and nerve fibers are still branching. "
Similarly, developing neurotransmitter and hormone cell receptors in the brain are vulnerable to disruption by neurotoxic agents. Exposure to drugs and chemical when neurotransmitter receptors are forming has been reported to affect the number of receptors and their functional maturation. Exposure to hormones can disrupt brain cell receptor development because certain receptors require reinforcement by the appropriate hormone at a critical stage in development. Many substances in the environment have hormone-like properties, including some chlorinated hydrocarbon pesticides.'9
Increased susceptibility of rodents to organophosphate pesticides appears to be due to insufficient detoxifying capabilities. For example, the greater toxicity of parathion and methyl parathion in the young has been shown to be a result of lower levels of detoxifying enzymes compared to the levels of enzymes that change, or activate, both organophosphates to their more potent neurotoxic metabolites (paraoxon and methyl paraoxon).'
Few studies on organophosphates have been conducted to determine the effects that low-level exposure during infancy has on neurological development. In fact, federal regulations currently do not require that any pesticides be evaluated for the effects of low-level exposure on behavior, including such processes as learning ability, activity level and memory, or on emotion, sight, and hearing. Organophosphates are the only class of pesticides that are currently required to undergo any neurotoxicity testing at all. This testing is limited solely to a screen to assess for delayed paralytic reactions following high level exposure.20
Although not required by federal regulation, some behavioral tests on developing animals have been conducted which indicate that low-level exposure to organophosphates and carbamates during nervous system development may impair neurological integrity . Studies suggest that exposure to certain organophosphates and carbamates before and immediately after birth can cause delays in reflex and sexual development, as well as delays in eye opening; alter nerve transmission function and neuroreceptor development, impair neuromuscular function, alter brain electrical activity, and, in several cases, affect brain structure.'6 2' 22
In fact, federal regulations currently do not require that any pesticide be evaluated for the effects of low-level exposure on behavior, including such processes as learning ability, activity level and memory, or on emotion, sight, and hearing."
While these studies do not answer the critical question of how long the sensitivity of the developing nervous system to organophosphates and carbamates might last, they collectively indicate that exposure to some organophosphates and carbamates early in neurological development may alter neurological function and may cause subtle and long-lasting neurobehavioral impairments.23

1. Natural Resources Defense Council. February, 1989. Intolerable risk: Pesticides in our children's food.
2. National Academy of Sciences. 1977. Drinking water and health, Vol. 1. Washington, DC: National Academy Press.
3. Calabrese EJ.1986 Age and susceptibility to toxic substances New York. John Wiley 7 Sons
4. World Health Organization (WHO) Environmental Criteria 59, Principles for evaluating health risks from chemicals during infancy and early childhood: The need for a special approach. Geneva, 1986.
5. Marquis, JK and GC Siek. 1988. Sensitive populations and risk assessment in environmental policy-making. In Saxena, J. (ed.), Hazard assessment of chemicals, Vol. 6. Washington, DC: Hemisphere Publishing Co.
6. Vessel, ES. 1983. Dynamically interacting genetic and environmental factors that affect the response of developing individuals to toxicants. In Hunt, VR, et al, (ed.), Banbury report 11: Environmental factors in human growth and development New York: Cold Spring Harbour Laboratory.
7. Murphy, SD. 1983. Toxicity and hepatic metabolism of organophosphate insecticides in developing rats. In Banbury Report 11 (see note 6).
8. Reiter, LW. 1986. Age-related effects of chemicals. In Banbury Report 11 (see note 6).
9. Mulvihill, JJ. 1982. Ecogenetic origins of cancer in the young: Environmental and genetic determinants. In Levine, AS, (ed.), Cancer in the young. New York: Marson Publishing.
10.Vesselinovitch, SD, et al. 1979. Neoplastic response of mouse tissues during perinatal age periods and its significance in chemical carcinogenesis. Perinatal Carcinogenesis. National Cancer Institute Monograph 51.
11. Dryoff, MC, et al. 1986. Correlation of 04 ethyideoxythymidine accumulation, hepatic initiation and heptacellular carcinoma induction in rats continuously administered diethyinitrosamine. Carcinogenesis 7:241-246.
12. Chang, MJ, et al 1981. Interrelationships between cellular proliferation, DNA alkylation and age as determinants of ethyinitrosourea induced neoplasia. Cancer Lett 13:39-45.
13. Chan, P, and TL Dao. 1983. Effects of dietary fat on age-dependent sensitivity to mammary carcinogenesis. Cancer Lett 18: 245-229.
14. Laib, RJ, et al 1985. The rat river foci bioassay: age-dependence of induction by vinyl chloride of ATP-deficient foci. Carcinogenesis 6:65-68.
15. Wiggins, RC. 1986. Myelination: A critical stage in development. Neurotoxicology 7(2): 103-129.
16. Spyker, JM, and David L Avery. 1977. Neurobehavioral effects of prenatal exposure to the organophosphate diazinon in mice. J. Toxicol and Environ. Health 3:989-1002.
17. Klaassen, DC, et al. 1986. Casarett and Doull's toxicology. New York: Macmillan.
18. Wester, RC, and Hl Maibach. 1982. Percutaneous absorbtion: Neonate compared to adult. In Banbury Report 11 (see note 6).
19. Kupfer, David. 1982. Studies on short and long-range estrogenic action of chlorinated hydrocarbon pesticides. In Banbury Report 11 (see note 6).
21. Mactutus, CF, and HA Tilson. 1986. Psychogenic and neurogenic abnormalities after perinatal insecticide exposure. In Riley, EP, and CV Vorhees (eds.), Handbook of behavioral teratology. New York: Plenum Press.
22. Rosenstein, L and N Chernoff. 1978. Spontaneous and evoked EEG changes in perinatal rats following in utero exposure to Baygon: A preliminary investigation. Bull Environ. Contam. Toxicol 20:624-632.
23. Calabrese, EJ. 1986. Age and susceptibility to 23. See also: National Academy of Sciences. 1985.
Citation for this article: Whyatt Robin 1989, "Intolerance risk : the physiological susceptibility of children to pesticides" Journal of Pesticide Reform Vol.9, No. 3, Fall 1989, pp 5 - 9
Copyright © 1989 Northwest Coalition for Alternatives to Pesticides.
Reprinted with permission.
Info Request | Services | Become EAP Member | Site Map
Give us your comments about the EAP site
Ecological Agriculture Projects, McGill University (Macdonald
Campus)
Ste-Anne-de-Bellevue, QC, H9X 3V9 Canada
Telephone:
(514)-398-7771
Fax:
(514)-398-7621
Email: info@eap.mcgill.ca
To report problems or otherwise comment on the structure of this site, send mail to the Webmaster